There’s a great paper from Pete Skene, postdoc 2011-2017 in the Henikoff lab, on eLife: An efficient targeted nuclease strategy for high-resolution mapping of DNA binding sites. The paper describes a novel method that performs as well as ChIP-Seq, but with lower background and false-positives. ChIP-seq is a widley used method and is used for transcription factor mapping at base-pair resolution, however it requires reasonable numbers of cells and there are issues with cross-linking artefacts, and limits on sensitivity.
The method is called CUT&RUN (cleavage under targets & release using nuclease). It uses unfixed nuclei which are attached them to a solid support; TF-antibody and pA-MN (MNase tagged with Protein A) are bound and MNase cleavage occurs on either side of the TF. MNase fragments (short) are released into the supernatant, while the genomic fragment (large) are retained in the unfixed nuclei. MNase fragments are available for standard library prep and sequencing but the paper shows that “CUT&RUN achieved base-pair resolution of mammalian TFs with only 10 million sequenced reads.”
Theres a description on the FredHutch website and a cpuple of figures from the paper here…enjoy!
Fig1: shows the outline of the method

Fig3: shows data from just 3 million reads per sample

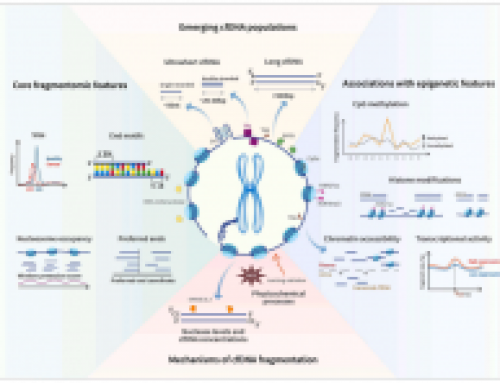






It is a great idea, but I doubt it will replace ChIP-seq because you can very easily mess it up by overdigesting (and people do mess it up that way all the time, and that issues exists to an extent even in the datasets in the paper).
P.S. One minor issue with your description:
“ChIP-seq is a widley used method and is used for transcription factor mapping at base-pair resolution”
We get base-pair resolution from ChIP-exo and ChIP-nexus. ChIP-seq is a bit more diffuse
Thanks for the comments Georgi. Your right about the base-pair resolution from ChIP-seq not being quite accurate. I’ve seen very little ChIP-exo but that’s probably because we’ve done so much standard ChIP there’s an inertia to moving to other methods.