Andrew Adey’s group at OHSU published sci-MET – a single cell method for whole-genome methylation analysis in Nature Biotech last month: Highly scalable generation of DNA methylation profiles in single cells. In this they described their indexing method and report on an analysis of over 3000 single-cells. They demonstrate the technology on cell lines and mouse cells. Methylation analysis allows us to determine where cancer is coming from in cell-free material, but only if we understand methylation in the tissue-of-origin (eg Methylation Deconvolution). This new method should allow researchers to build much better methylation maps for more sensitive diagnostics test (see MIMIC). See also the NIH Directors blog and Epigenie (who map out the protocol).

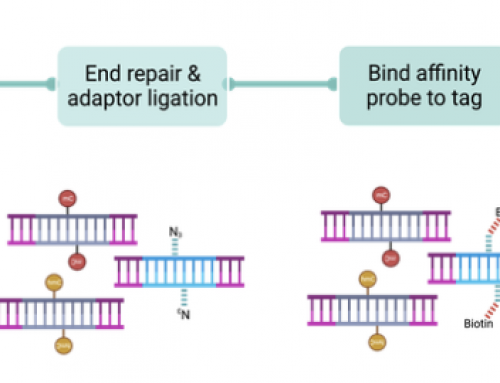
sci-MET works by first adding a unique DNA indexes to each cell (molecular tagging is accomplished with barcoded Tn5 transposases), the DNA library is then bisulfite converted before amplification and sequencing (Fig1a opposite). Cells are identified from the two 10-nt indexes introduced by PCR, and the 11-nt index introduced by tagmentation. Because cells are combined for bisulfite conversion and sequencing the costs drop precipitously over previous approaches, and linking back to individual cells allows identification of the various cell types in a tissue.

To demonstrate how well the sci-MET method works the team analysed 708 single-cells from B-lymphoblast cell line (GM12878) with low depth sequencing (55,000 unique reads per cell) and produced “methylation profiles that closely matched expectation for the GM12878 cell line”. They also analysed a mix of three human cell lines, which were processed and correctly sorted by methylation pattern. In the paper they also describe sequencing of 606 single-cells from an in vivo mouse model revealing a distinct enrichment for each neuronal cluster within sets of excitatory and inhibitory differentially methylated regions (DMRs).
The team have shown they can profile thousands of cells simultaneously. Adey said. “This technology reduces the cost to prepare single-cell DNA methylation libraries to less than 50 cents per cell from $20 to $50 per cell.”






Leave A Comment